Clinical Trials
Download Brochure





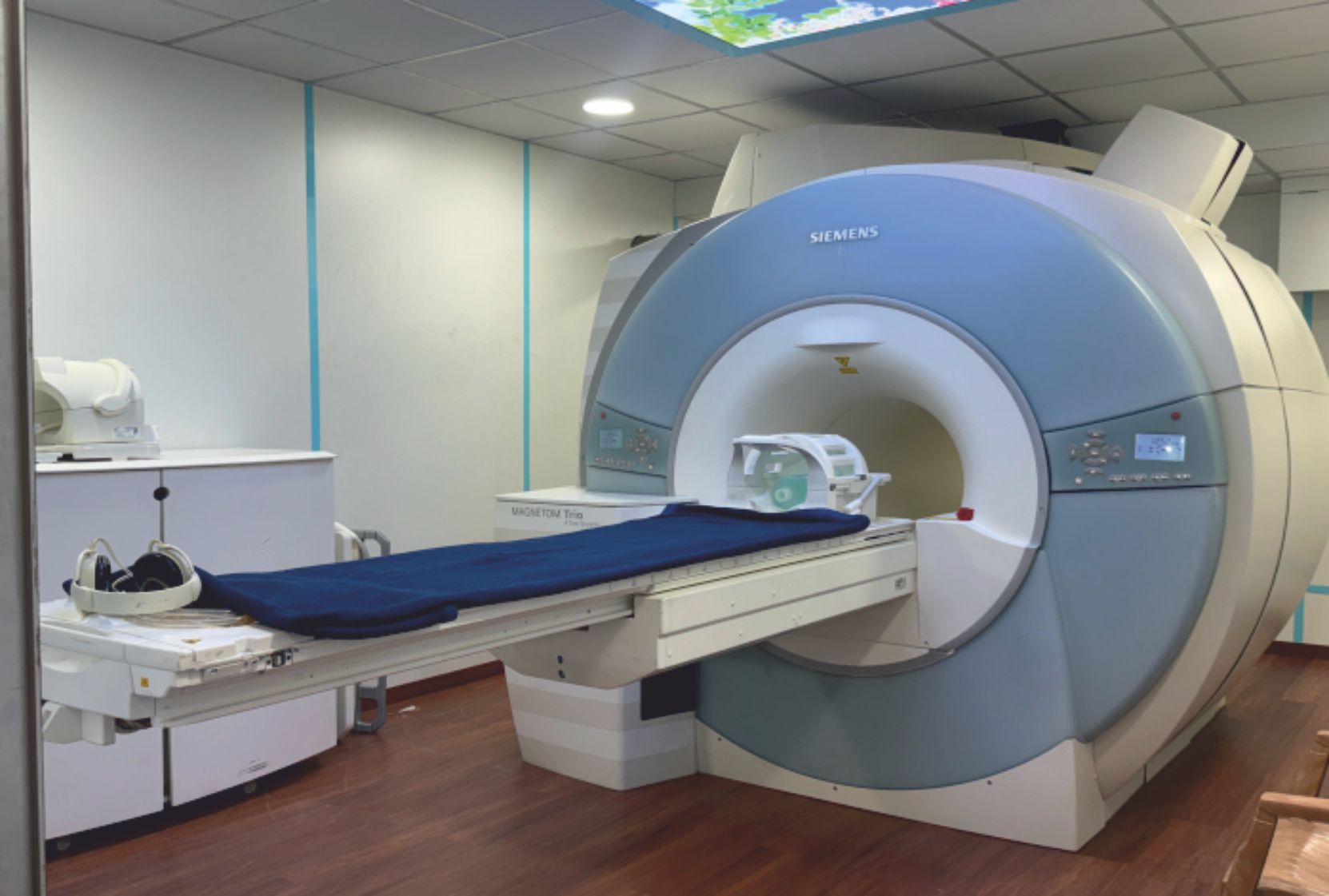
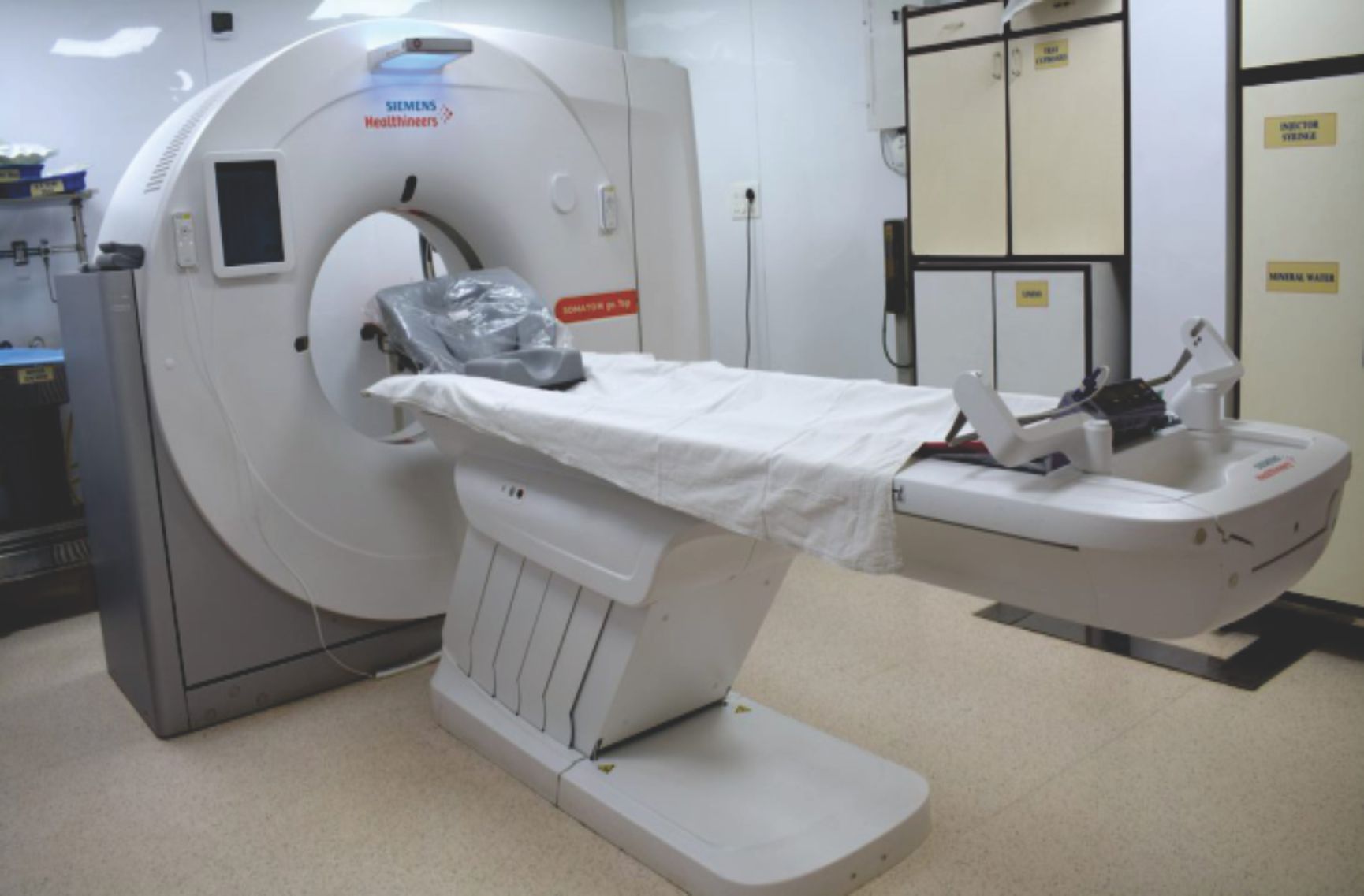

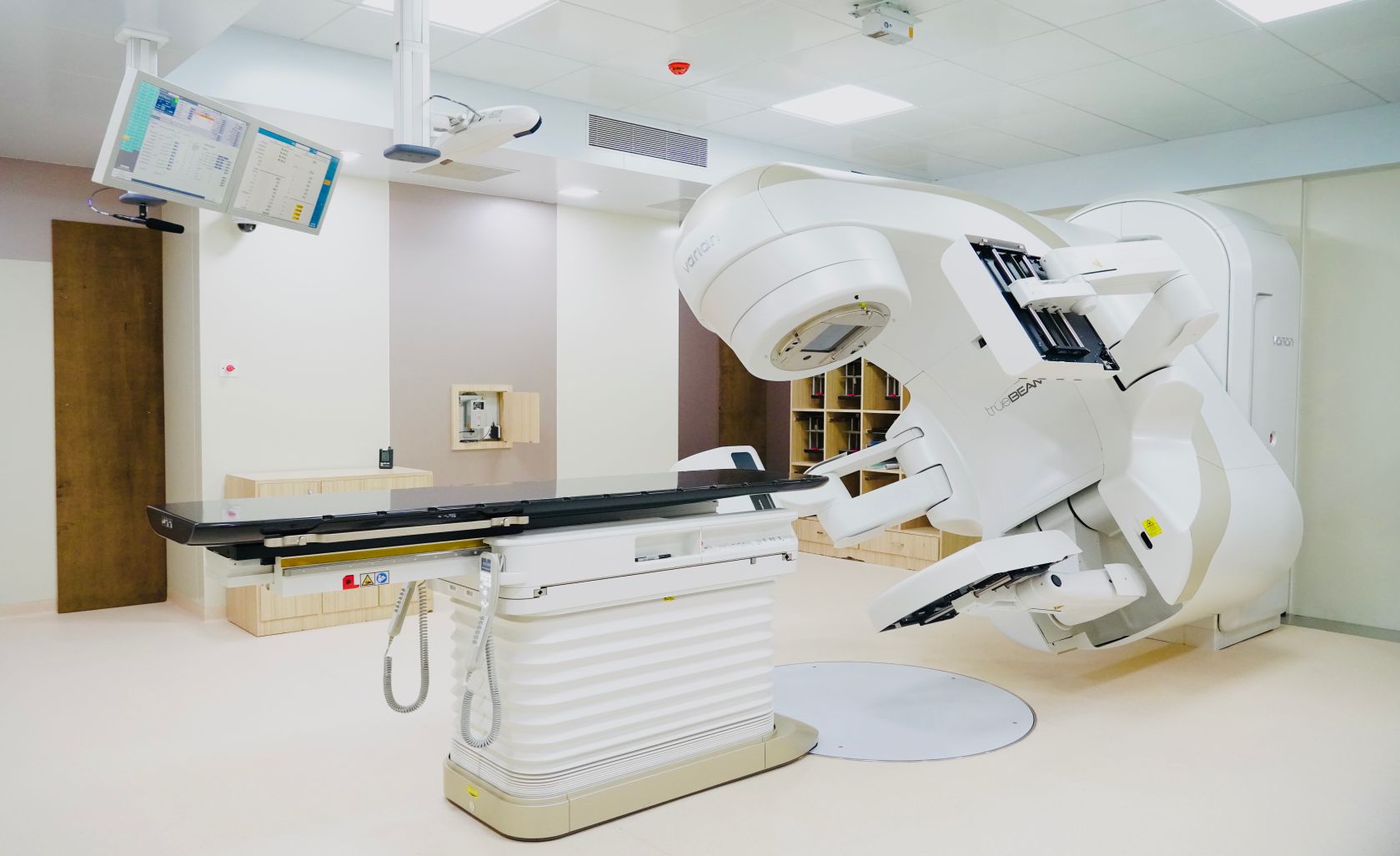
About Clinical Research Department
The Clinical Research Department at Bhaktivedanta Hospital and Research Institute is a state-of-the-art research facility committed to promoting scientific, ethical, and high-quality clinical research. The department collaborates with national and international organizations to advance healthcare, innovation, and patient outcomes.
Location:3rd Floor, B-wing, Vrindavan building.
Hospital and Infrastructure Facilities

Protocol Development & Scientific Publications Unit
- Dedicated unit for hospital-initiated (investigator-driven) studies
- Expertise in protocol design, development, and regulatory compliance
- End-to-end support: concept → protocol → ethics approval → study execution
- Team of clinical researchers, biostatisticians, and medical writers
- Expertise in manuscript writing and publication strategy
- Enhances global visibility and academic impact of research
Monitoring Room
- Dedicated space for sponsor and CRO monitors
- Comfortable, quiet, and fully equipped workstations
- Secure and confidential environment for document review
- On-site support for document coordination and queries
- Efficient handling of multiple concurrent monitoring visits
- Designed to ensure smooth, compliant, and timely monitoring



Secure Study Document Storage Facility
- Dedicated storage area for all study-related documents
- Locked cupboards with access limited to authorized study personnel
- Clearly labelled and organized storage system for easy retrieval
- Ensures confidentiality and data security
- Efficient management of multiple concurrent studies
- Maintains document integrity throughout the study lifecycle

Authorized Department Access
- Robust access control system ensuring secure and compliant research environment
- Restricted entry to authorized personnel only
- Protection of sensitive study data, documents, and resources
- Enhances data integrity and risk management
- Ensures adherence to regulatory and ethical standards
- Controlled and coordinated access for sponsors, monitors, and auditors
- Maintains confidentiality and smooth operational workflow
Centrifuge & Blood Sample Processing Unit
- Dedicated in-house centrifuge and sample processing facility
- Ensures timely and efficient sample handling
- Maintains sample integrity and accuracy
- Operated by trained and qualified staff
- Supports multiple concurrent clinical studies
- Ensures proper documentation and quality control


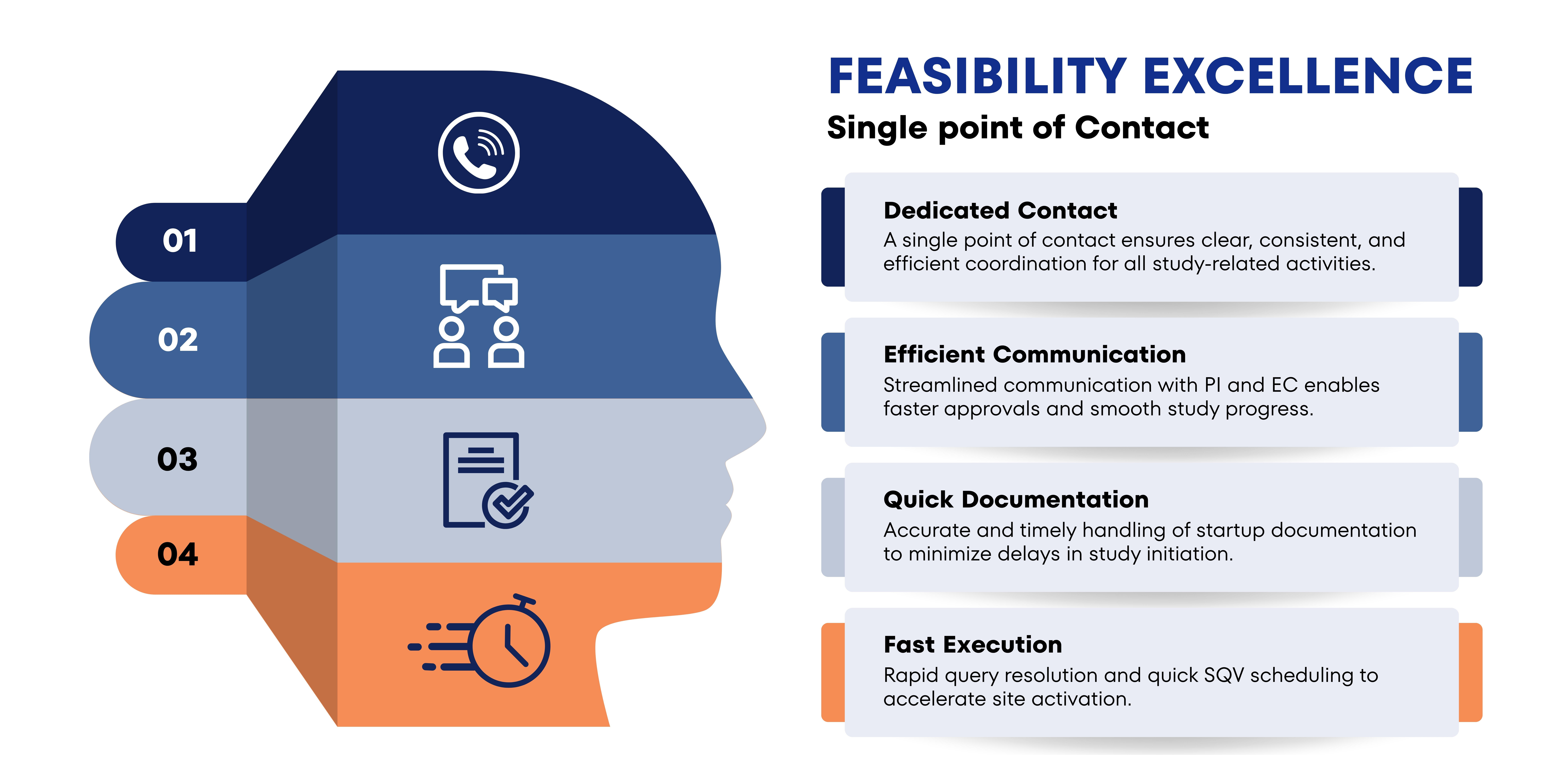
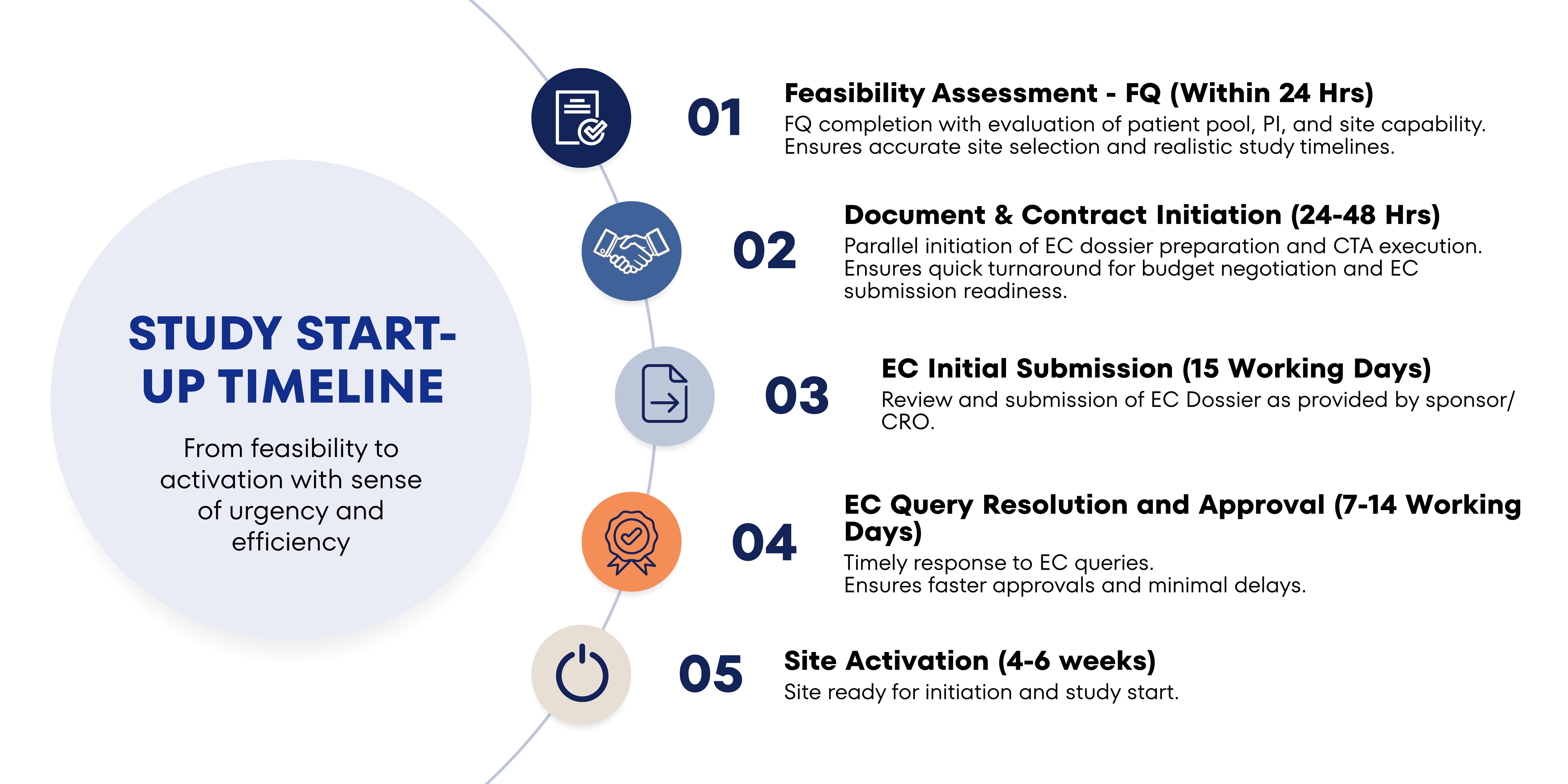
Dedicated Feasibility & Start-Up Team
- Dedicated feasibility team for new study assessments
- Single point of contact for sponsors and CROs
- Strong focus on quick response and efficient coordination
- Ensures fast start-up timelines
- Proactive approach with a sense of urgency
- Streamlined communication and decision-making process
- Supports smooth and accelerated study initiation
Patient Room
- Dedicated patient room within the department
- Designed for investigator-initiated studies
- Comfortable space for patient rest and observation
- Equipped for vital signs monitoring and basic assessments
- Ensures patient safety, privacy, and convenience
- Supports efficient study conduct and patient management
- Maintains a calm and patient-friendly environment

INTERNATIONAL

DOMESTIC
International Clinical Trial Department & Domestic Offices
- Dedicated workspace for Clinical Research Coordinators (CRCs)
- Supports management of national and international clinical trials
- Promotes efficient coordination and documentation
- Equipped for data entry, reporting, and communication
- Encourages team collaboration and workflow efficiency
- Ensures adherence to protocols and regulatory requirements
- Facilitates timely and accurate study execution



Investigational Product (IP) Storage Facility
- Controlled access to IP storage area
- Temperature-controlled refrigerators for product stability
- Dedicated study-specific refrigerators
- Refrigerators with different temperatures (25–30°C, 2–8°C, −20°C, and −70°C)
- Lock-and-key system for enhanced security
- Ensures proper segregation and traceability
- Maintains product integrity and compliance
- Supports regulatory requirements and audit readiness
Ethics Committee Office

- Dedicated Ethics Committee (EC) office
- Ensures efficient and timely review of study protocols
- Maintains confidentiality and secure documentation
- Supports submission, review, and approval processes
- Facilitates communication between investigators and EC
- Ensures adherence to regulatory and ethical guidelines
- Enhances audit readiness and compliance
Access-Controlled Data Centre Unit

- Access-controlled data centre unit
- Ensures secure storage of clinical trial data
- Restricted access to authorized personnel only
- Maintains data confidentiality and integrity
- Supports data backup and secure management systems
- Complies with regulatory and data protection standards
- Enhances audit readiness and data reliability

Virtual Tour & Insights
Institute virtual tour and infrastructure
Scientific & Industrial Research Organization
CONTACT
US
Dr. Vijaykumar Gawali (MBBS, D-Stat, PCPV, PGCR)
Head – Clinical Research & Academics | Medical Superintendent – Centres of Excellence at Bhaktivedanta Hospital & Research Institute, Mumbai
NABH Assessor – EC & Clinical Trials Accreditation
Dr. Tejasvee Kadam (BDS)
Lead – Clinical Research at Bhaktivedanta Hospital & Reserach Institute

