Clinical Research Course

.JPG)
.JPG)
.jpg)


































































Quick Enquiry
Fill in your details and we'll get back to you
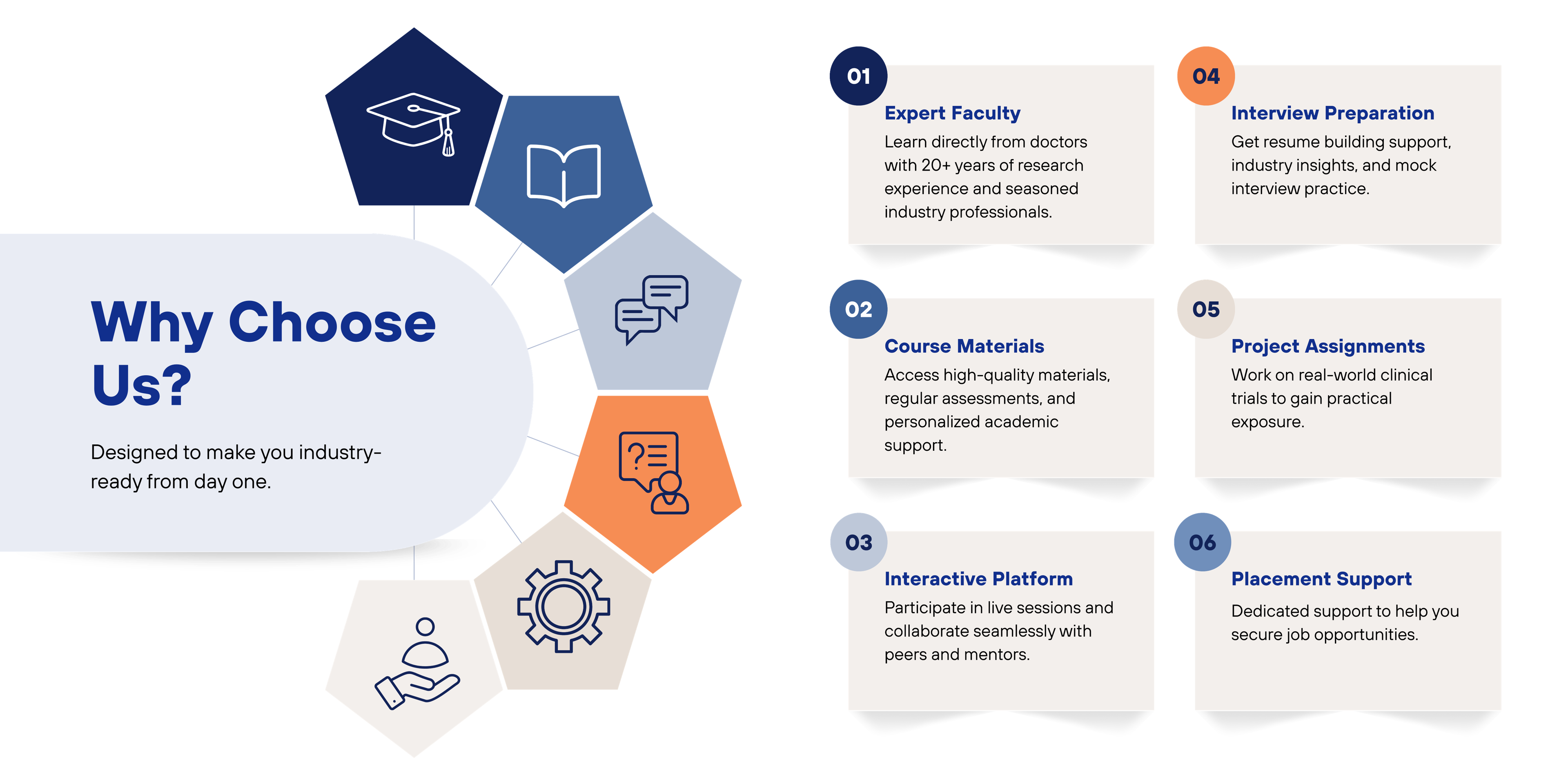
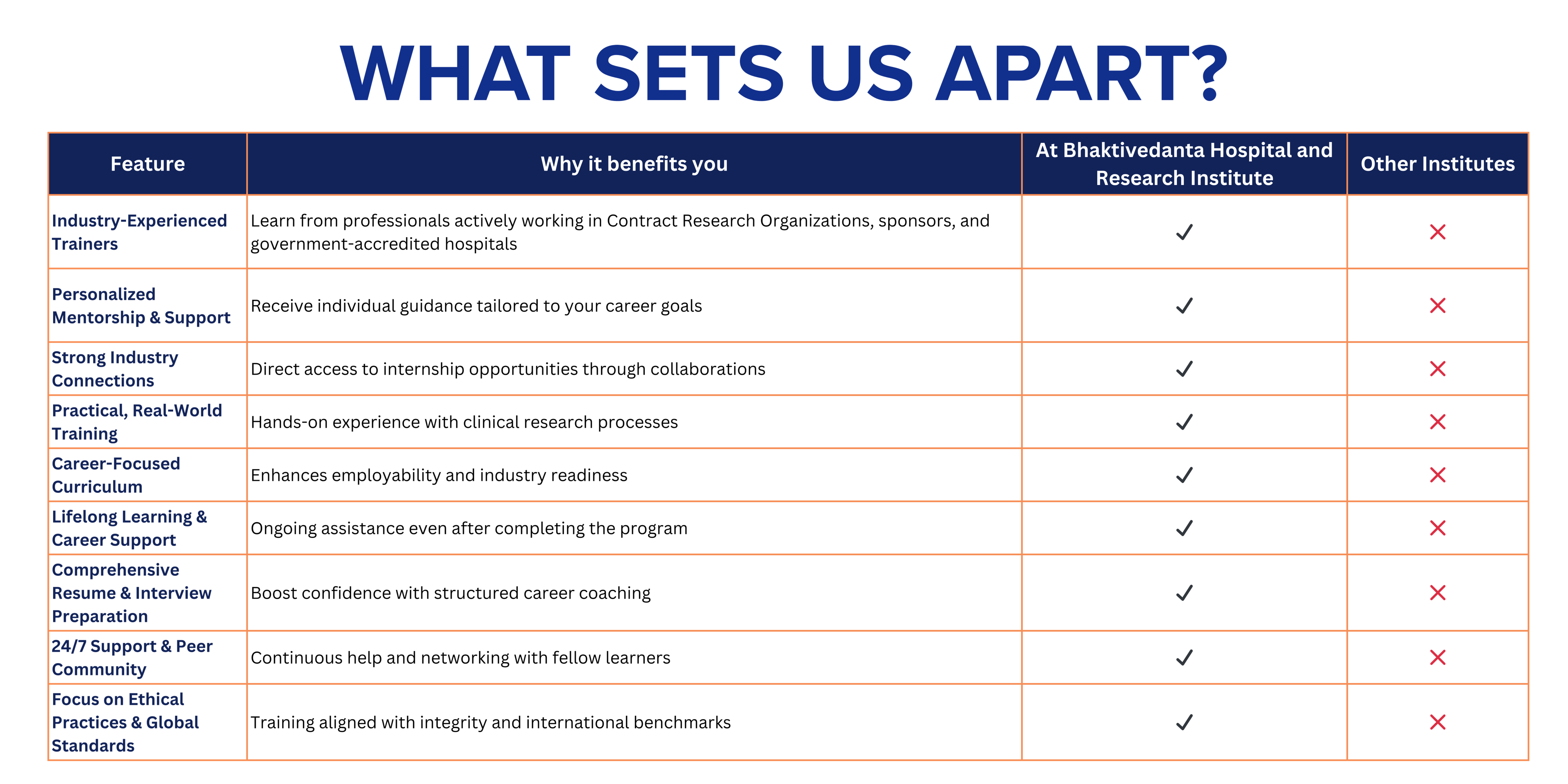
The Post Graduate Clinical Research (PGCR) course is a 15-month comprehensive program that integrates strong theoretical foundations with extensive hands-on clinical research training. The course commences in July and concludes in September of the following year, ensuring in-depth exposure to real-world clinical research practices.
Admissions for the PGCR course open in February each year and follow a simple three-step selection process. Applicants receive the selection intimation within 7 working days after completion of the interview.
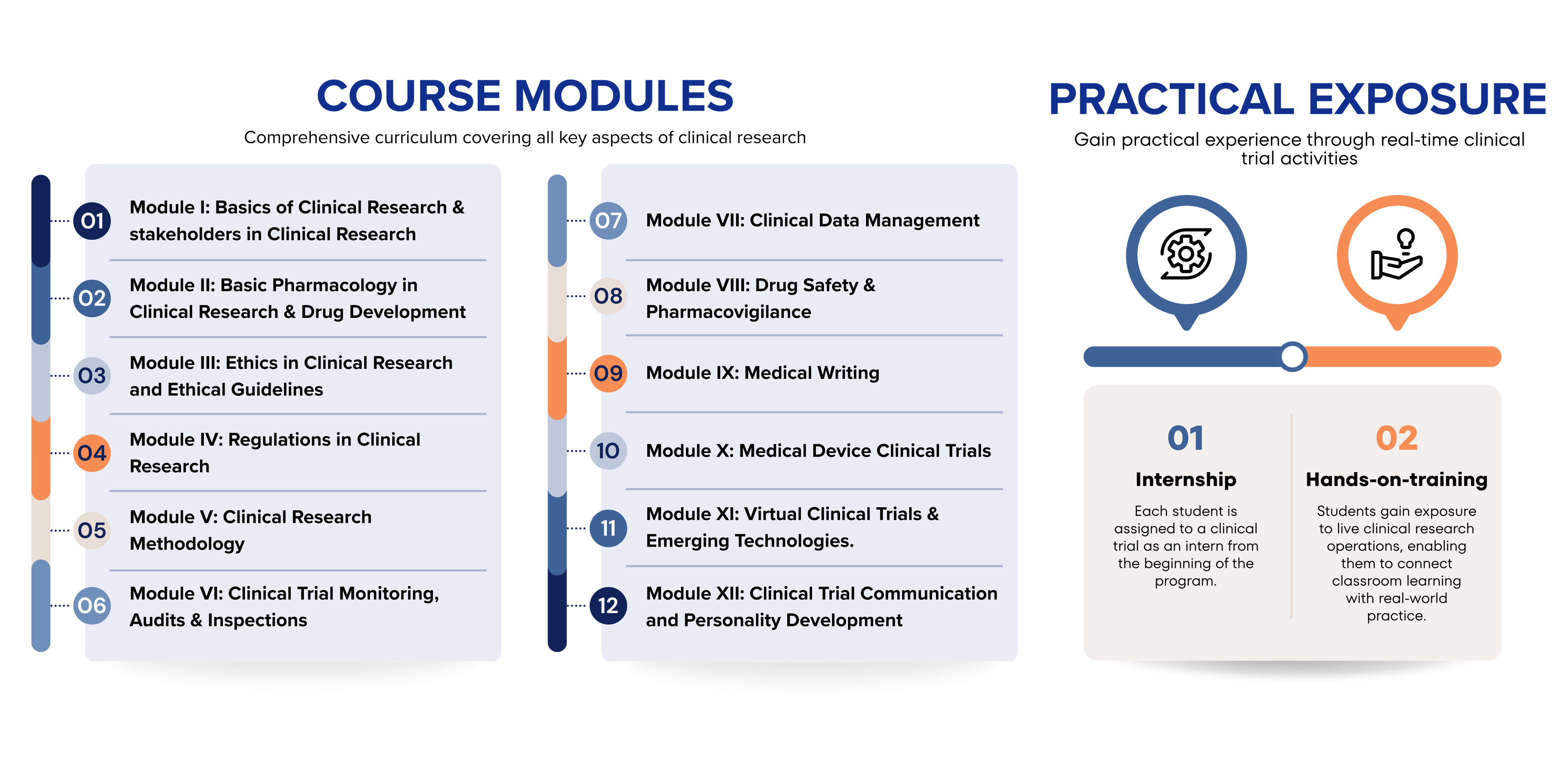
The curriculum comprises 11 structured academic modules along with one year of intensive hands-on training, designed to equip students with practical skills and regulatory knowledge essential for a successful career in clinical research.

CONTACT
US
Dr. Vijaykumar Gawali (MBBS, D-Stat, PCPV, PGCR)
Head – Clinical Research & Academics | Medical Superintendent – Centres of Excellence at Bhaktivedanta Hospital & Research Institute, Mumbai
NABH Assessor – EC & Clinical Trials Accreditation
Dr. Tejasvee Kadam (BDS)
Lead – Clinical Research at Bhaktivedanta Hospital & Reserach Institute

